New approach methodologies (NAMs) are used in research as potential alternatives to animals and are progressively reducing and replacing animal use when feasible. Examples include human cell-based studies, which have been used in research since the early 1950s, and more cutting-edge methods such as patient-derived organoids, organ-on-a-chip and computer modelling.
Why are NAMs used in research?
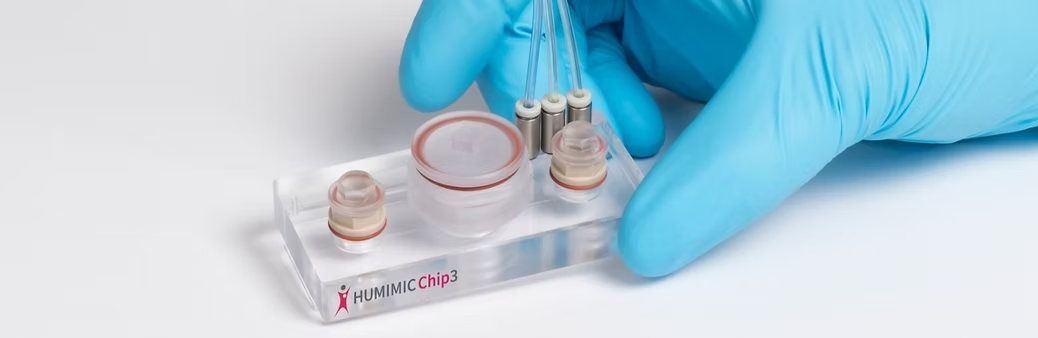
Are NAMs ready to replace all animal use in research?
Which types of NAMs are used in research?
Whether human, animal or engineered, cells that are grown in the laboratory and maintained (described as cultured) outside a living organism, have been used within science for generations. The first human cell line was obtained in 1951, during the treatment of a patient with cancer, Henrietta Lacks. These cells, called HeLa, have contributed to many research advances. Cells of interest can be extracted from living tissue to be examined more closely under carefully controlled conditions, allowing researchers to understand how tissue grows, study specific mechanisms within the cells, test the effect of substances on cells, generate pharmaceuticals based on living cells and organisms, and much more.
The fact that cells are grown in a very controlled environment offers advantages in terms of reproducibility and consistency, which allow for precise quantification and validation of scientific findings. Cells are also cost-effective and a way to study human or animal health without ethical constraints.
When it comes to regulatory testing, lab-based (in vitro) methods are already well-established and validated in assessing certain types of toxicity, such as for the skin, to test sensitivity and irritation. Here, different compounds that are found in common products, that may trigger an allergic reaction for instance, can be used to cause such a reaction in human tissue models, to understand the effect it will have.
However, there are some key limitations of using in vitro, the major one being tied to the very nature of these techniques, as cells in a dish are unlikely to behave in exactly the same way as in a living organism or their natural environment. Cells are also grown flat in a dish, whereas in the body they are surrounded by other cells in a 3D arrangement, and they interact with their environment, engaging in complex communication with their surroundings and other cells and tissues.
In which research areas are NAMs more widely used?
NAMs are also used for greater scientific understanding (basic research) and translating basic research to develop new drugs and treatments. Through a combination of techniques and information, gleaned from both animal and non-animal studies, researchers can build the best possible understanding.
NAMs are limited in the complexity of information they can provide about the living body, but often they can model more faithfully certain aspects of some isolated human biological systems, closed mechanisms and predict interactions, helping to improve the translation of research findings from the lab to the clinic.
Artificial intelligence can simulate some biological processes or even how certain diseases progress, leading to the identification of possible drug compounds, as well as predictions about how they might behave once in the body. A study led by the Spanish National Research Council trained an AI system to detect brainwaves in monkeys that are disrupted in neurological disorders such as Alzheimer’s, based on brain recordings from mice. Without artificial intelligence these brainwaves could otherwise be missed by standard imaging techniques, contributing to the improvement of the diagnosis of brain diseases.
Meanwhile, cells grown in the lab – from human cells to more complex organoids and organs-on-chips – have been developed for a range of different diseases and conditions, including neurodegenerative diseases and cancer, and are showing promise for studying disease mechanisms, drug responses and developing personalised medicines – and in some cases reducing or replacing the use of animals.
In a study at the University of Manchester, UK, researchers used lung organoids, produced from human stem cells, to investigate the possible effects of carbon-based nanomaterials on human health, with the results mirroring the negative effects on lung health shown in animal studies. Alternatives have also been used in basic research, such as in work at the University of Birmingham, also in the UK, that developed an organ-on-a-chip which replicated the blood vessels in the human liver, allowing scientists to understand how immune cells reach liver cells.
NAMs, while they do not reach the full maturation of an organ inside a living organism, provide an important window into early developmental stages. By trying to replicate the development of an organ in a controlled, simpler system, scientists can pinpoint the steps involved in organ development and they can understand how certain substances affect it. Because these models are often human-based, they can capture mechanisms that are specific to humans. In 2025, researchers from IBEC, Spain, using a platform that mimicked the wall of an uterus, were able to capture the first video of a human embryo implantation, which could help improve understanding of the natural process and improve fertility and assisted reproduction techniques.